Abstract
Background: Cold agglutinin disease is an autoimmune hemolytic anemia with limited treatment options and no established standard of care. The pathophysiology is driven by the classical complement pathway in which IgM auto-antibodies bind erythrocytes and fix complement via initial binding and activation of the C1 complex generating active C1s protease. Anemia results from extravascular hemolysis of complement opsonized erythrocytes, primarily in the liver. The anti-C1s antibody BIVV009 inhibits C1s activity, and specifically blocks the classical complement pathway, leaving the alternate and lectin complement pathways intact. We hypothesized that classical complement pathway blockade using BIVV009 would prevent hemolysis, correct anemia, and obviate the need for transfusions in patients with primary cold agglutinin disease.
Methods: Six patients primary cold agglutinin disease patients were enrolled in an open label Ph1/1b trial. The study was conducted in three parts: Part A, single ascending doses in healthy volunteers (HV); Part B, multiple ascending doses in HV; and Part C, multiple doses in patients with four classical complement mediated diseases including cold agglutinin disease. Patients in Part C received a test dose of 10 mg/kg BIVV009, followed by a full dose of 60 mg/kg 1-4 days later, and three additional weekly doses of 60 mg/kg. Biweekly fixed doses of 5.5g were used for maintenance therapy in a subsequent Named Patient Program.
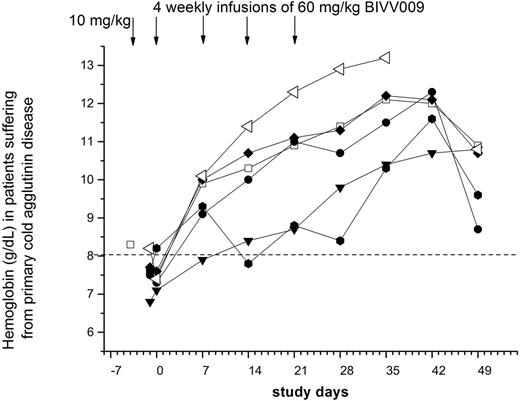
Results: All infusions were well tolerated without need for pre-medication, and pharmacokinetic data demonstrated that BIVV009 infusions supported biweekly treatment. BIVV009 concentrations >18µg/mL inhibited the classical pathway of complement activation (as assessed by the Wieslab-CP assay). BIVV009 infusion subsequently raised endogenous C4 levels 3.2-fold (95%CI: 2.4-4.0 fold; p<0.05) demonstrating classical complement inhibition and reduced complement factor consumption. In addition, deposition of C3d on erythrocytes decreased as measured by flow cytometric analysis, consistent with evidence of classical complement pathway inhibition and reduced erythrocyte opsonization. BIVV009 rapidly abrogated extravascular hemolysis, as measured by normalizing bilirubin levels within 24 hours, normalized haptoglobin levels in 4 of 6 patients within one week, and restoring absolute reticulocyte counts to levels appropriate for the degree of anemia. All 6 of 6 primary cold agglutinin disease patients responded with a hemoglobin increase >3.5 g/dL (mean 4.3g/dl; 95%CI: 3.8-4.9 g/dL; individual best response; p<0.05) within 6 weeks; the lower 95% confidence interval for the responder rate was 54%. Hemolysis and anemia recurred when effective drug levels were cleared from the circulation approximately 3-4 weeks after the last dose of BIVV009 for all patients. Re-exposure to BIVV009 in the named patient program recapitulated the efficacy, again in all patients. Maintenance therapy demonstrated persistent responses without related adverse effects or development of clinically relevant neutralizing antibodies, now with up to 18 months of observation. All five transfusion-dependent patients became and remained transfusion-free during treatment.
Conclusion: BIVV009 was well tolerated and immediately stopped hemolysis in 6 of 6 severely anemic patients suffering from primary cold agglutinin disease, increasing hemoglobin by a mean of 4.3g/dl and precluding the need for transfusions while on BIVV009. Based on these encouraging results, BIVV009 received FDA Breakthrough Therapy designation and pivotal trials will be initiated to further define BIVV009 safety and efficacy profiles.
Figure 1: individual hemoglobin response patterns in primary cold agglutinin disease patients during the initial exposure of 6 weeks
Jaeger: Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis Pharmaceuticals Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses. D'Sa: Janssen Cilag: Consultancy, Honoraria, Other: Education grant, Research Funding; Amgen: Consultancy, Honoraria, Research Funding. Patou: Inc., a Bioverativ Inc. Company: Employment. Panicker: Bioverativ, Inc.: Employment, Equity Ownership. Parry: Bioverativ Therapeutics Inc.: Employment. Gilbert: TrueNorth Therapeutics Inc., a Bioverativ Inc. Company: Other: formerly Employment. Jilma: Biomed: Research Funding; Recardio: Research Funding; Emcools: Research Funding; Bayer: Research Funding; Themis: Research Funding; Syntheract: Research Funding; Bioverativ: Research Funding; TrueNorth Therapeutics: Research Funding; JHL: Research Funding; Baxalta: Research Funding; Arsanis: Research Funding; VitaerisBios: Research Funding; TrueNorth Therapeutics Inc.: Consultancy; Valneva: Research Funding; Prediction Biosciences: Research Funding; Boehringer Ingelheim: Research Funding; Octapharma: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal